Recombinant Human IGF-II R/IGF2R protein (rFc Tag)
Species
Human
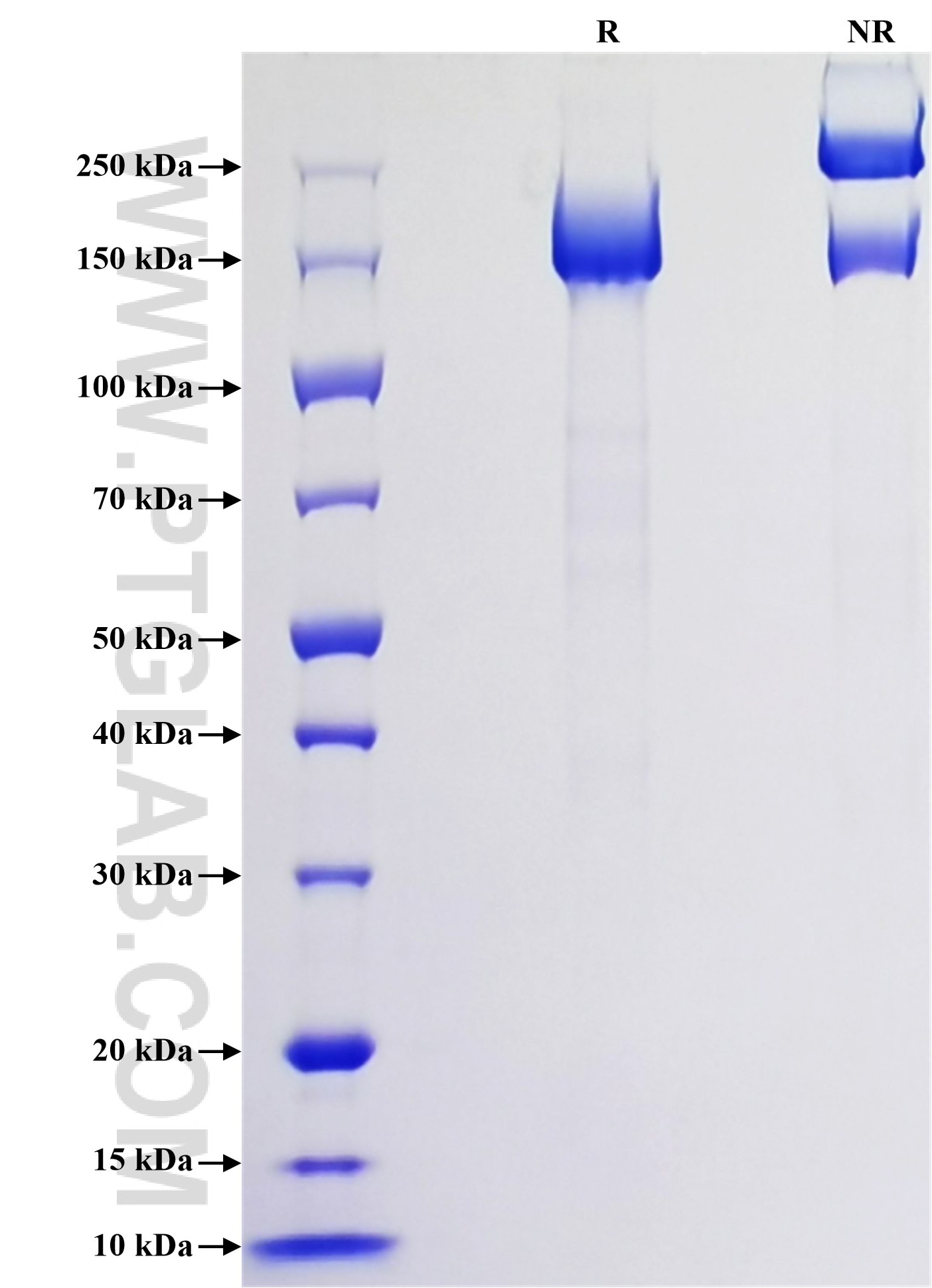
Purity
>90 %, SDS-PAGE
Tag
rFc Tag
Activity
not tested
Cat no : Eg3488
Validation Data Gallery
Product Information
| Purity | >90 %, SDS-PAGE |
| Endotoxin | <0.1 EU/μg protein, LAL method |
| Activity |
Not tested |
| Expression | HEK293-derived Human IGF-II R protein Gln41-Asp1365 (Accession# P11717) with a rabbit IgG Fc tag at the C-terminus. |
| GeneID | 3482 |
| Accession | P11717 |
| PredictedSize | 172.5 kDa |
| SDS-PAGE | 140-230 kDa, reducing (R) conditions |
| Formulation | Lyophilized from 0.22 μm filtered solution in PBS, pH 7.4. Normally 5% trehalose and 5% mannitol are added as protectants before lyophilization. |
| Reconstitution | Briefly centrifuge the tube before opening. Reconstitute at 0.1-0.5 mg/mL in sterile water. |
| Storage Conditions |
It is recommended that the protein be aliquoted for optimal storage. Avoid repeated freeze-thaw cycles.
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the recommended temperature. |
Background
IGF-II R (insulin-like growth factor 2 receptor), also known as IGF2R or M6P/IGF2R, is a multifunctional type I transmembrane glycoprotein. Its primary function is to act as a "scavenger receptor," binding and internalizing ligands such as insulin-like growth factor-II (IGF-II) and lysosomal enzymes marked with mannose-6-phosphate, thereby mediating their lysosomal degradation and negatively regulating cell growth, proliferation, and metabolism. This receptor plays a crucial role in embryonic development, tumor suppression, and metabolic homeostasis. Unlike IGF1R, its cytoplasmic tail is short and lacks tyrosine kinase activity. It primarily inhibits pro-growth signals by clearing circulating IGF-II through an endocytosis-degradation pathway, functioning as a tumor-suppressive "scavenger." As a classic tumor suppressor, its gene deletion or functional inactivation is commonly observed in various cancers, leading to abnormal activation of pro-growth signaling pathways.
References:
1. Shirakawa, Jun et al. Diabetes vol. 74,12 (2025): 2351-2364. 2. Bella, Pamela et al. EMBO molecular medicine vol. 12,1 (2020): e11019. 3. Guo, Xiaorong et al. International journal of biological macromolecules vol. 284,Pt 1 (2025): 137785. 4. Han, Jie et al. Autophagy vol. 17,6 (2021): 1393-1409.