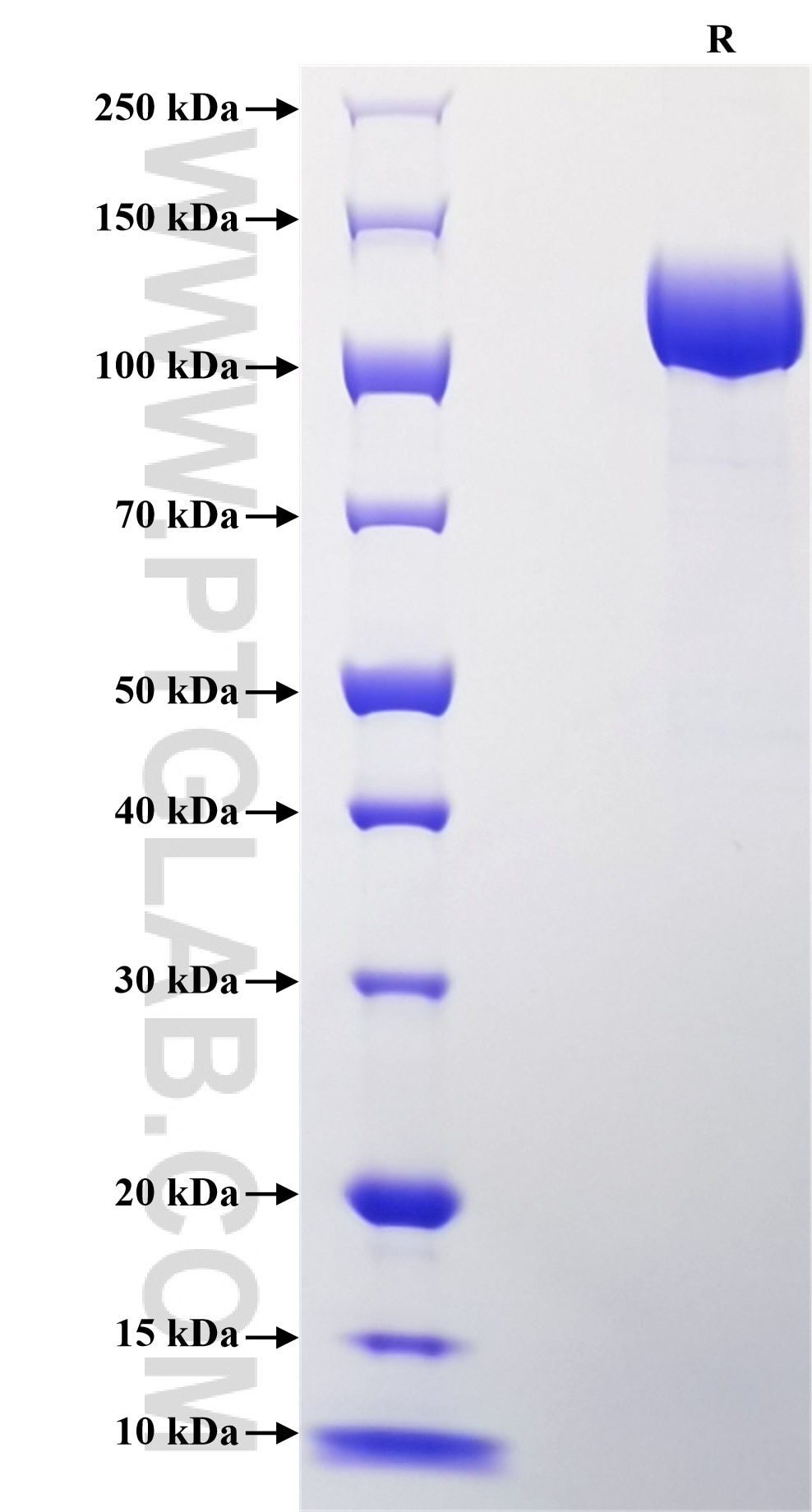
Recombinant Human AGRN protein (rFc Tag)
Species
Human
Purity
>90 %, SDS-PAGE
Tag
rFc Tag
Activity
not tested
Cat no : Eg5123
Validation Data Gallery
Product Information
| Purity | >90 %, SDS-PAGE |
| Endotoxin | <0.1 EU/μg protein, LAL method |
| Activity |
Not tested |
| Expression | HEK293-derived Human AGRN protein Ala1260-Pro2045 (Accession# O00468-6) with a rabbit IgG Fc tag at the N-terminus. |
| GeneID | 375790 |
| Accession | O00468-6 |
| PredictedSize | 109.5 kDa |
| SDS-PAGE | 100-140 kDa, reducing (R) conditions |
| Formulation | Lyophilized from 0.22 μm filtered solution in PBS, pH 7.4. Normally 5% trehalose and 5% mannitol are added as protectants before lyophilization. |
| Reconstitution | Briefly centrifuge the tube before opening. Reconstitute at 0.1-0.5 mg/mL in sterile water. |
| Storage Conditions |
It is recommended that the protein be aliquoted for optimal storage. Avoid repeated freeze-thaw cycles.
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the recommended temperature. |
Background
Agrin, a large heparan sulfate proteoglycan, is expressed in neuronal, as well as nonneuronal tissues. Nerve-derived agrin has been demonstrated to play an essential role in development and maintenance of the neuromuscular junction. The agrin gene encodes a protein of more than 2000 amino acids with predicted molecular weight of 225 kDa but can display as 600 kDa protein due to extensive N-terminal glycosylation. Neurotrypsin cleaves the proteoglycan agrin at two homologous, highly conserved sites, liberating a middle 90-kDa and a C-terminal 22-kDa fragment from the membrane or ECM-bound N-terminal moiety of agrin.
References:
1. Bezakova, Gabriela, and Markus A Ruegg. Nature reviews. Molecular cell biology vol. 4,4 (2003): 295-308. 2. Kummer, Terrance T et al. Current opinion in neurobiology vol. 16,1 (2006): 74-82. 3. Stephan, Alexander et al. FASEB journal : official publication of the Federation of American Societies for Experimental Biology vol. 22,6 (2008): 1861-73.