- HEK293 expressed
- Endotoxin-free
- Animal-component free
HumanKine® recombinant human Sonic Hedgehog (SHH) protein
Cat no : HZ-1306
Synonyms
HHG 1, HHG1, HLP3, HPE3, MCOPCB5, SHH, SMMCI, Sonic hedgehog protein, TPT, TPTPS
Validation Data Gallery
Technical Specifications
| GeneID | 6469 |
| Species | Human |
| Expression | HEK293 |
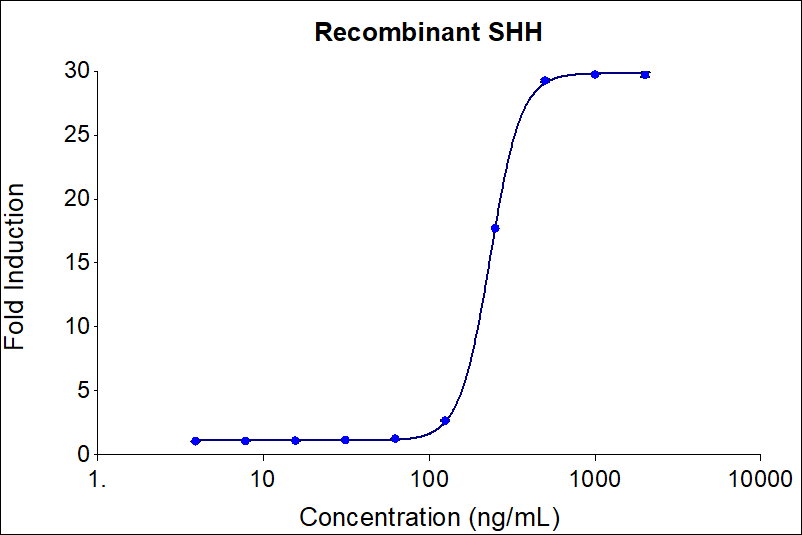
| Activity | Typically ≤350 ng/mL |
| Purity | >90% |
| Endotoxin | <1.0 EU/μg |
| Accession Number | Q15465 |
| Molecular Mass | 19-22 kDa reduced and non-reduced, monomer, glycosylated |
| Formulation | 10mM Tris pH 7.4 + 100mM NaCl + 1% CHAPS, See Certificate of Analysis for details |
| Species Reactivity | human,mouse |
Stability and Reconstitution
| Stability and Storage | Product Form | Temperature Conditions | Storage Time (From Date of Receipt) |
|---|---|---|---|
| Lyophilized | -20°C to -80°C | Until Expiry Date | |
| Lyophilized | Room Temperature | 2 weeks | |
| Reconstituted as per CofA | -20°C to -80°C | 6 months | |
| Reconstituted as per CofA | 4°C | 1 week | |
| Avoid repeated freeze-thaw cycles. | |||
| Reconstitution | Briefly centrifuge the vial before opening. It is recommended to reconstitute the protein to 0.1 mg/mL in sterile water containing 0.1% endotoxin-free recombinant human serum albumin (HSA). Gently swirl or tap vial to mix. |
Background
SHH, also named as HHG-1, belongs to the hedgehog family. SHH binds to the patched receptor (PTC), that in association with smoothened (SMO) activate the transcription of target genes. In the absence of SHH, PTC represses the constitutive signaling activity of SMO. SHH is synthesized as a 45 kDa protein precursor that is autocatalytically cleaved to yield of 20 kDa N-terminal fragment (24-197 in the human gene sequence) knowing as being responsible for all known hedgehog biological activity and a 25 kDa C-terminal fragment that contains the auto-processing machinery (PMID:10753901).
Synonyms
HHG 1, HHG1, HLP3, HPE3, MCOPCB5, SHH, SMMCI, Sonic hedgehog protein, TPT, TPTPS
Publications
| Species | Title |
|---|---|
Mol Biol Rep Discovery of YS-1 as a cell line of gastric inflammatory cancer-associated fibroblasts | |
EBioMedicine Leukemia inhibitory factor is a therapeutic target for renal interstitial fibrosis |