Human uPAR/PLAUR ELISA Kit
Sensitivity
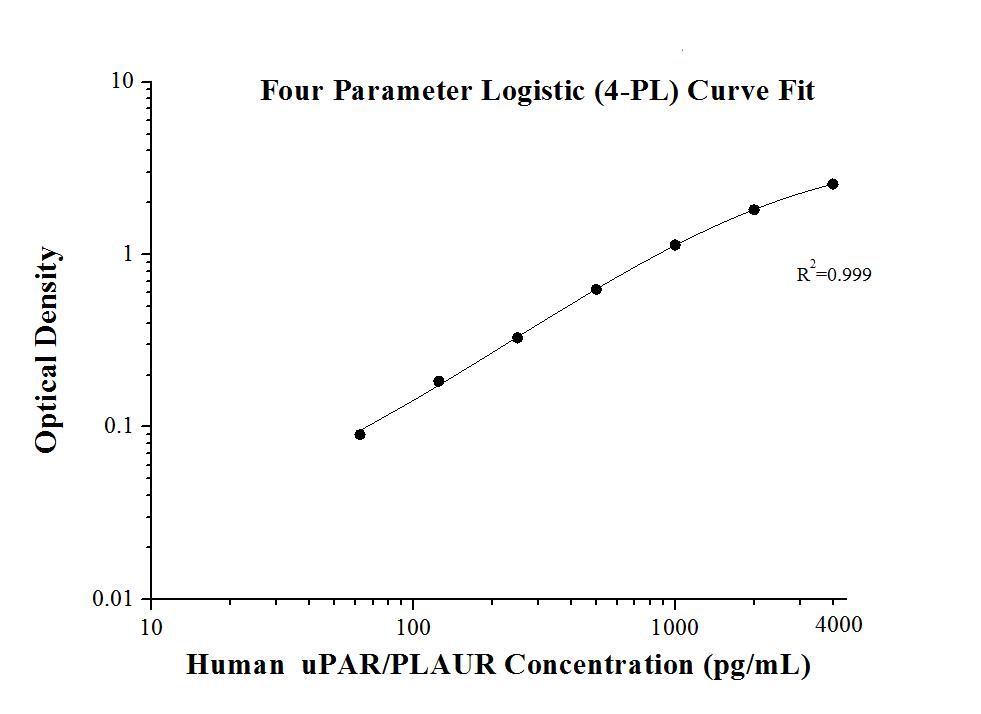
7.0 pg/mL
Range
62.5-4000 pg/mL
Reactivity
Human
Cat no : KE00206
Synonyms
CD87, MO3, PLAUR, U PAR, UPAR, URKR
Validation Data Gallery
Product Information
KE00206 is a solid phase sandwich Enzyme Linked-Immuno-Sorbent Assay (Sandwich ELISA). The uPAR/PLAUR ELISA kit is to be used to detect and quantify protein levels of endogenous uPAR/PLAUR. The assay recognizes human uPAR/PLAUR. An antibody specific for uPAR/PLAUR has been pre-coated onto the microwells. The uPAR/PLAUR protein in samples is captured by the coated antibody after incubation. Following extensive washing, another antibody of biotinylated specific for uPAR/PLAUR is added to detect the captured uPAR/PLAUR protein.For signal development, Streptavidin-HRP is added, followed by Tetramethyl-benzidine (TMB) reagent. Solution containing sulfuric acid is used to stop color development and the color intensity which is proportional to the quantity of bound protein is measurable at 450 nm with the correction wavelength set at 630 nm .
| Product name | Human uPAR/PLAUR ELISA Kit |
| Tests | 1 X 96 well plate |
| Sample type | Serum, Plasma, Cell culture supernatants, Urine |
| Assay type | Sandwich |
| Sensitivity | 7.0 pg/mL |
| Range | 62.5-4000 pg/mL |
| Reactivity | Human |
| Tested applications | Sandwich ELISA |
| Gene ID (NCBI) | 5329 |
Recovery
| Sample Type | Average | Range |
|---|---|---|
| Human serum | 89% | 76%-112% |
| Cell culture supernatants | 105% | 82%-119% |
| Urine | 90% | 77%-111% |
IntraAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 20 | 256.3 | 12.5 | 4.9 |
| 2 | 20 | 512.2 | 16.3 | 3.2 |
| 3 | 20 | 2,009.1 | 49.8 | 2.5 |
InterAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 24 | 259.7 | 21.6 | 8.3 |
| 2 | 24 | 501.1 | 28.5 | 5.7 |
| 3 | 24 | 1,863.5 | 154.6 | 8.3 |
Background Information
uPAR/PLAUR is a highly glycosylated GPI-anchored membrane protein. In addition to the membrane-anchored form, uPAR is released from the plasma membrane by cleavage of the GPI anchor and can be found as a soluble form (suPAR). uPAR contains three homologous domains (D1-D3) of which the N-terminal one (D1) represents the uPA-binding domain. After binding to uPAR, uPA cleaves plasminogen, generating the active protease plasmin which is involved in a wide variety of physiologic and pathologic processes. In addition to regulating proteolysis, uPAR has important function in cell adhesion, migration and proliferation. Studies reveal that uPAR expression is elevated during inflammation and tissue remodelling and in many human cancers, in which it frequently indicates poor prognosis. suPAR has been detected in plasma, and increased plasma concentrations of suPAR have been found in patients with some advanced cancers.
Properties
| Storage Instructions | All the reagents are stored at 2-8℃ for 6 months or -20℃ for 12 months. Refer to the protocol for further storage instructions. |
| Synonyms | CD87, MO3, PLAUR, U PAR, UPAR, URKR |