Human gp130 ELISA Kit
Sensitivity
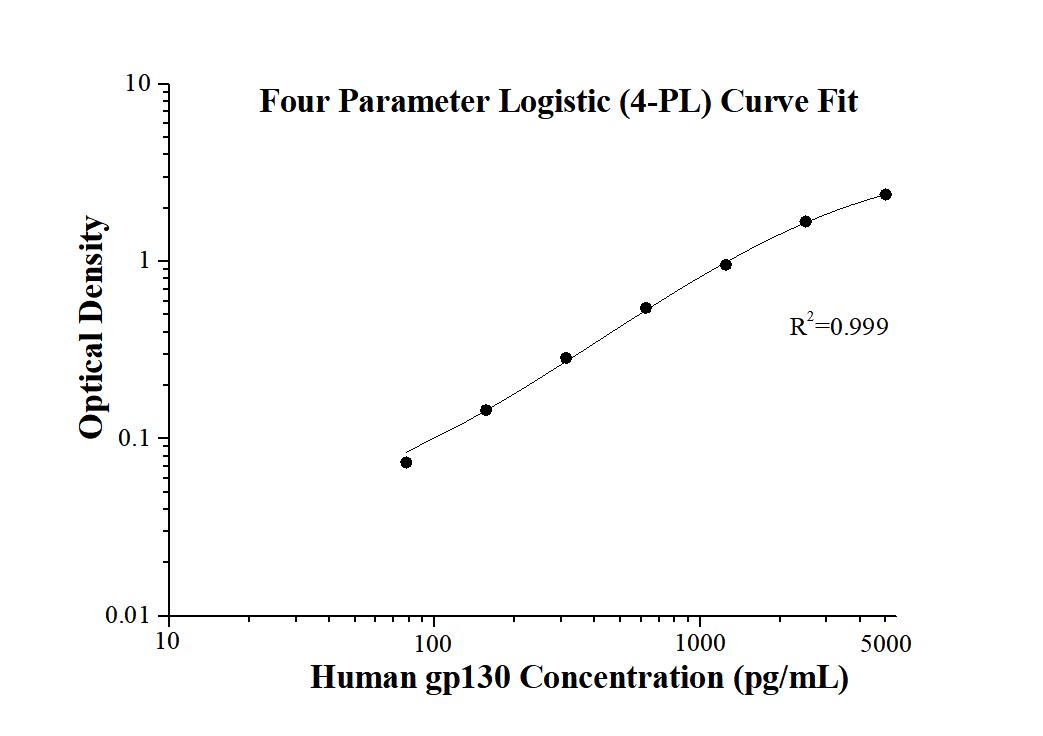
1.8 pg/mL
Range
78.1-5000 pg/mL
Reactivity
Human
Cat no : KE00176
Synonyms
IL6ST, CD130, Glycoprotein 130, GP130 RAPS, IL 6 receptor subunit beta
Validation Data Gallery
Product Information
KE00176 is a solid phase sandwich Enzyme Linked-Immuno-Sorbent Assay (Sandwich ELISA). The gp130 ELISA kit is to be used to detect and quantify protein levels of endogenous gp130. The gp130 ELISA kit is designed to measure human soluble gp130 in serum, plasma and cell culture supernatant. The assay recognizes human gp130. An antibody specific for gp130 has been pre-coated onto the microwells. The gp130 protein in samples is captured by the coated antibody after incubation. Following extensive washing, another antibody of biotinylated specific for human gp130 is added to detect the captured human gp130 protein. For signal development, Streptavidin-HRP is added, followed by Tetramethyl-benzidine (TMB) reagent. Solution containing sulfuric acid is used to stop color development and the color intensity which is proportional to the quantity of bound protein is measurable at 450 nm with the correction wavelength set at 630 nm.
| Product name | Human gp130 ELISA Kit |
| Tests | 1 X 96 well plate |
| Sample type | Serum, Plasma, Cell culture supernatants |
| Assay type | Sandwich |
| Sensitivity | 1.8 pg/mL |
| Range | 78.1-5000 pg/mL |
| Reactivity | Human |
| Tested applications | Sandwich ELISA |
| Gene ID (NCBI) | 3572 |
Recovery
| Sample Type | Average | Range |
|---|---|---|
| Human serum | 89% | 78%-98% |
| Cell culture supernatants | 101% | 93%-110% |
IntraAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 20 | 146.2 | 3.1 | 2.1 |
| 2 | 20 | 575.3 | 2.6 | 2.6 |
| 3 | 20 | 2,397.1 | 3.3 | 3.3 |
InterAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 24 | 149.5 | 6.1 | 4.1 |
| 2 | 24 | 577.7 | 16.4 | 2.8 |
| 3 | 24 | 2,270.1 | 88.3 | 3.9 |
Background Information
Glycoprotein 130 (gp130, also known as IL6ST, CD130 or IL6-beta) is a ubiquitously expressed, signal-transducing receptor that serves as the signal transduction unit for IL-6 family of cytokines, including IL-6, IL-11, IL-27, leukemia inhibitory factor (LIF), OSM, ciliary neurotrophic factor (CNTF), cardiotrophin 1 (CT-1), and cardiotrophin-like cytokine (CLC). These cytokines signal through the gp130/Jak/STAT pathway. Binding of IL-6 to IL-6R induces gp130 homodimerization and formation of a high-affinity receptor complex, which activates Jaks. That causes phosphorylation of gp130 tyrosine residues which in turn activates STAT3. gp130 is a type I transmembrane protein, and can also exist as a soluble form (sgp130). sgp130 binds to sIL-6R/IL-6 complexes and prevents their interactions with membrane-anchored gp130 on target cells.
Properties
| Storage Instructions | All the reagents are stored at 2-8℃ for 6 months or -20℃ for 12 months. Refer to the protocol for further storage instructions. |
| Synonyms | IL6ST, CD130, Glycoprotein 130, GP130 RAPS, IL 6 receptor subunit beta |