Validation Data Gallery
Tested Applications
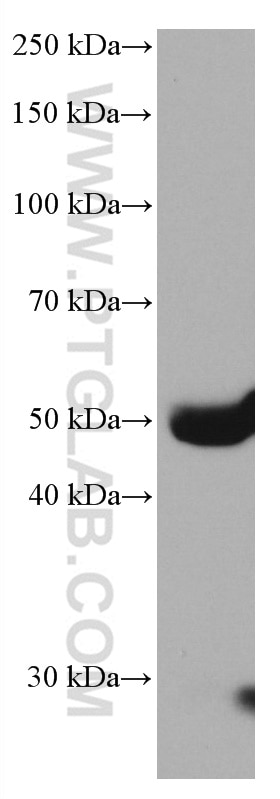
| Positive WB detected in | human plasma tissue |
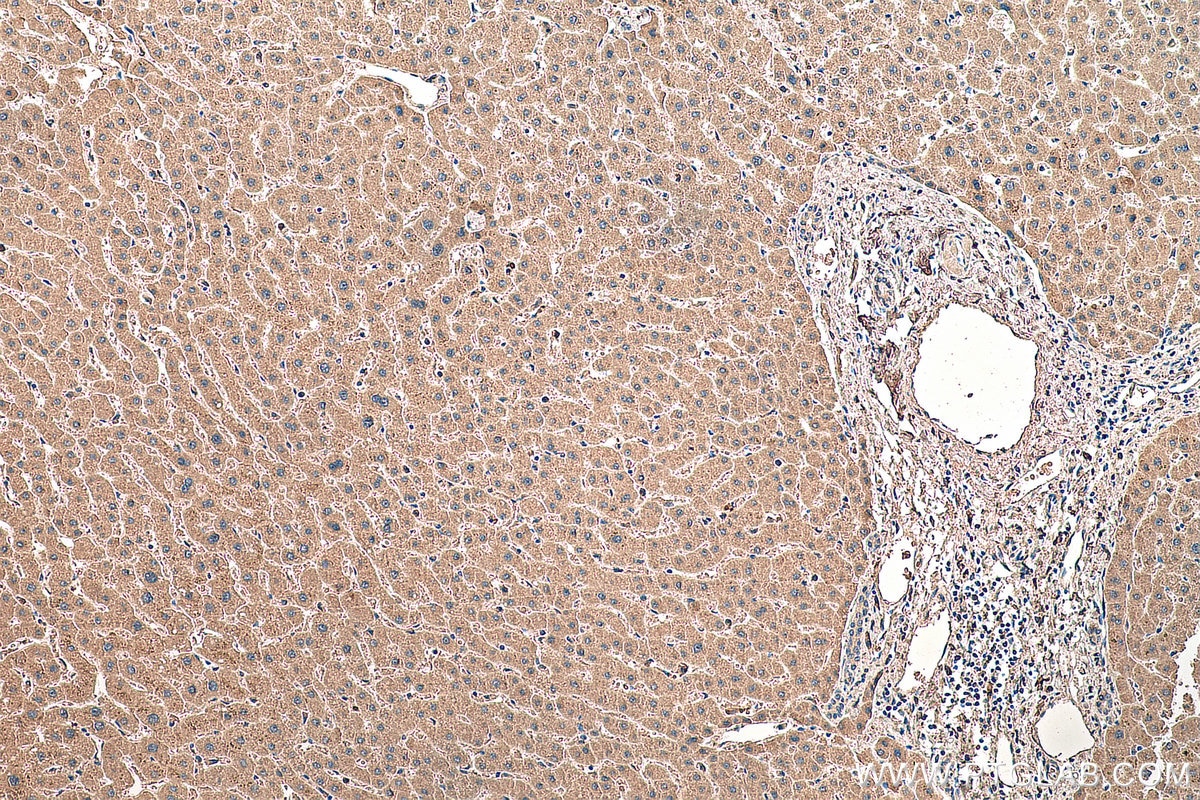
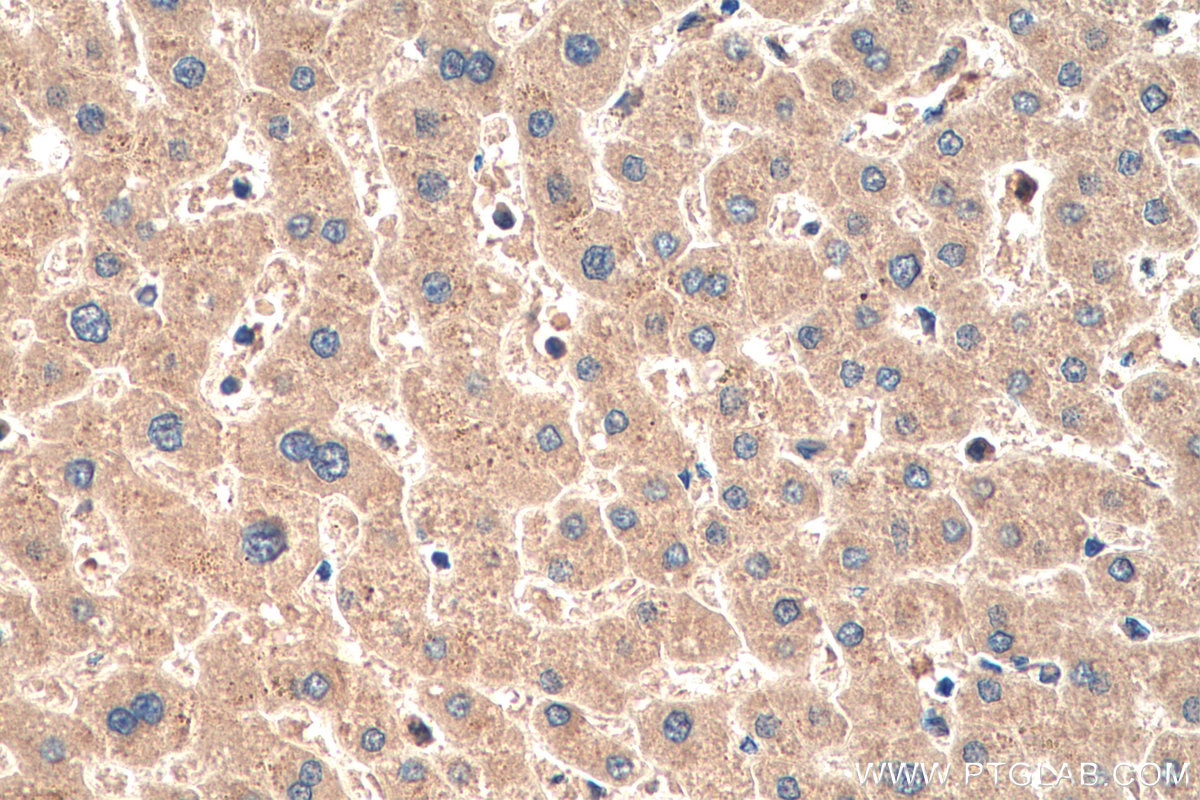
| Positive IHC detected in | human liver tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
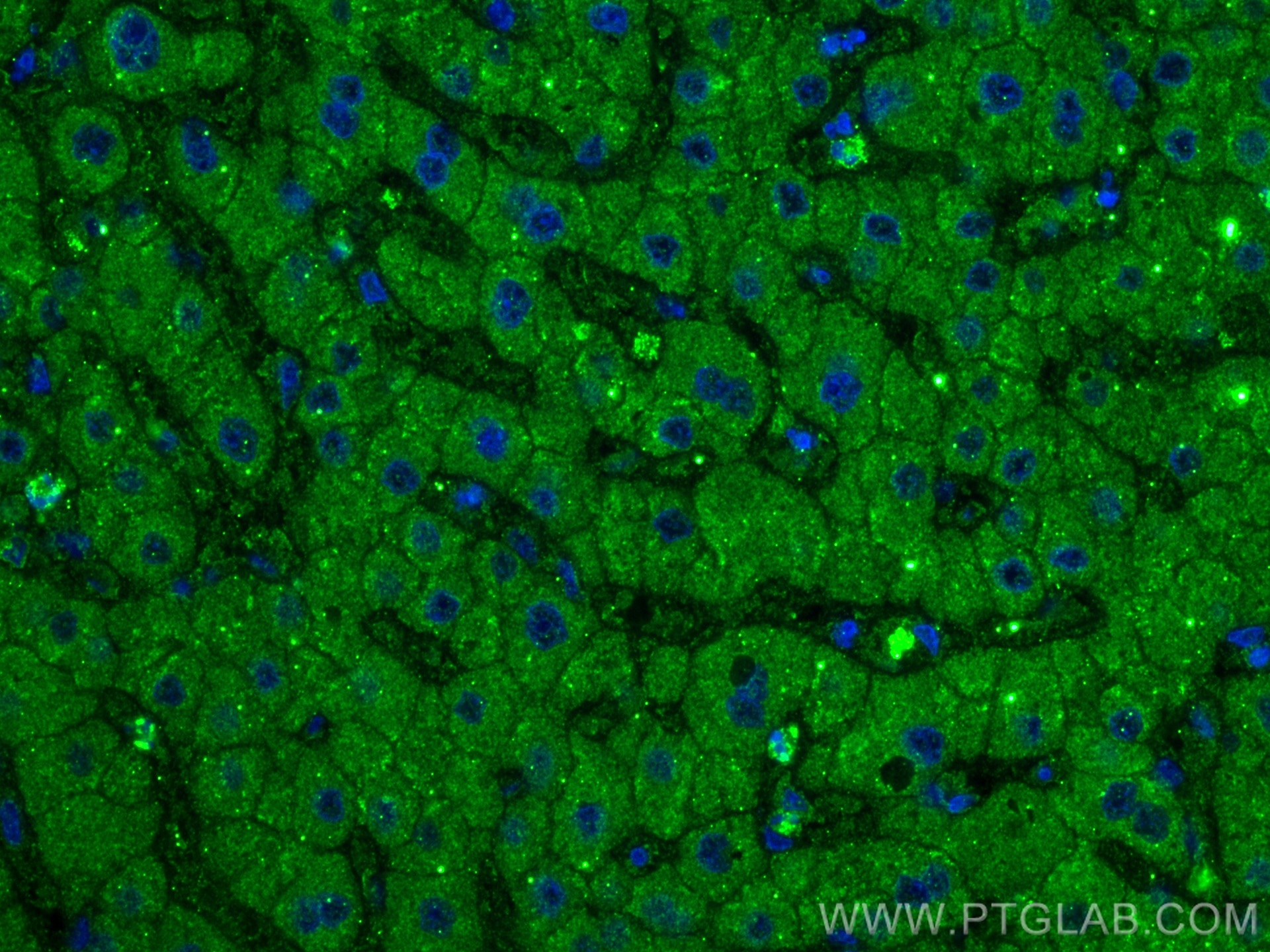
| Positive IF-P detected in | human liver tissue |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:5000-1:50000 |
| Immunohistochemistry (IHC) | IHC : 1:500-1:2000 |
| Immunofluorescence (IF)-P | IF-P : 1:200-1:800 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Product Information
66753-1-Ig targets Factor X in WB, IHC, IF-P, ELISA applications and shows reactivity with human samples.
| Tested Reactivity | human |
| Host / Isotype | Mouse / IgG1 |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag11664 Product name: Recombinant human F10 protein Source: e coli.-derived, PET28a Tag: 6*His Domain: 140-488 aa of BC046125 Sequence: CHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERRKRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQECKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGEAVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGIVSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSGGPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPEVITSSPLK 相同性解析による交差性が予測される生物種 |
| Full Name | coagulation factor X |
| Calculated molecular weight | 488 aa, 55 kDa |
| Observed molecular weight | 50 kDa |
| GenBank accession number | BC046125 |
| Gene Symbol | F10 |
| Gene ID (NCBI) | 2159 |
| RRID | AB_2882099 |
| Conjugate | Unconjugated |
| Form | |
| Form | Liquid |
| Purification Method | Protein G purification |
| UNIPROT ID | P00742 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol{{ptg:BufferTemp}}7.3 |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. |
Background Information
Coagulation factor X (F10) is a vitamin K-dependent glycoprotein that participates in the middle phase of the blood coagulation cascade (PMID: 1931959; 1313796). It is synthesized as a single-chain precursor in the liver where it undergoes a number of covalent processing steps before secretion into the blood as a two-chain molecule linked by a disulfide bond (PMID: 1313796). The light chain contains 2 EGF-like domains, while the heavy chain contains the catalytic domain which is structurally homologous to those of the other hemostatic serine proteases. Factor X is activated into factor Xa, by both the extrinsic and intrinsic pathway. The activated factor then converts prothrombin to thrombin in the presence of factor Va, calcium and phospholipid during blood clotting.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for Factor X antibody 66753-1-Ig | Download protocol |
| IHC protocol for Factor X antibody 66753-1-Ig | Download protocol |
| WB protocol for Factor X antibody 66753-1-Ig | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |